Exploring Dental Implant Clinical Trials in Italy
For individuals residing in Italy who are considering dental implants, an option to evaluate is the participation in dental implant clinical trials. These trials are designed to assess new techniques and technologies related to dental implants. Individuals interested in participating must meet specific eligibility criteria that typically include factors such as age, oral health status, and previous dental treatments. Understanding the potential benefits and considerations of joining a clinical trial can provide valuable insights for making informed decisions.
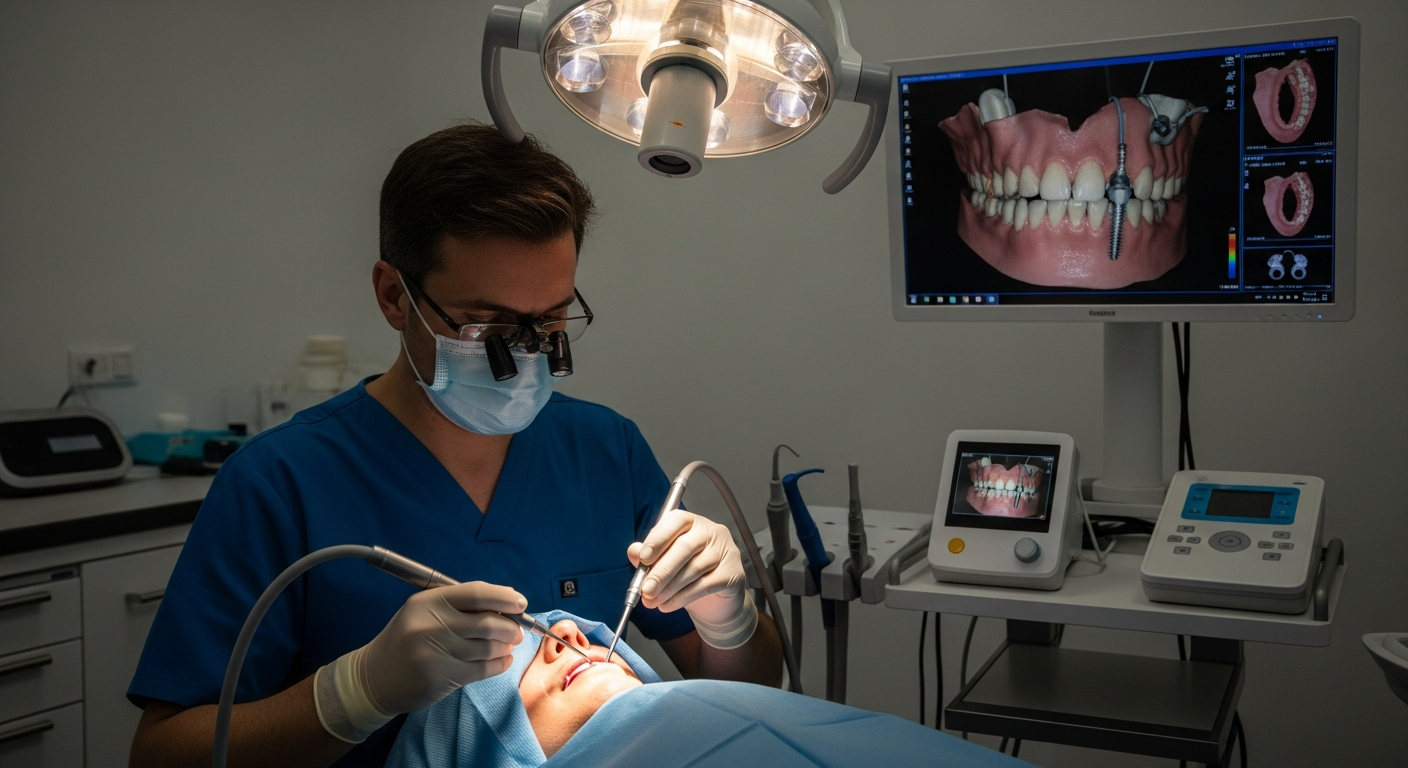
Clinical trials represent a vital component of medical and dental research, allowing researchers to test new procedures, materials, and treatment approaches before they become widely available. In Italy, dental implant clinical trials provide patients with opportunities to receive cutting-edge care while helping to shape the future of restorative dentistry. These trials are conducted under strict ethical guidelines and regulatory oversight to ensure participant safety and scientific integrity.
Understanding the Process of Dental Implant Clinical Trials in Italy
Dental implant clinical trials in Italy follow a structured process designed to evaluate the safety and effectiveness of new implant systems, surgical techniques, or materials. The process typically begins with preclinical research, followed by several phases of human trials. Phase I trials assess safety in a small group, Phase II evaluates effectiveness and side effects in a larger population, and Phase III compares the new treatment against standard care in extensive studies. Italian research institutions and dental clinics collaborate with regulatory bodies such as the Italian Medicines Agency (AIFA) to ensure compliance with European Union standards. Participants undergo thorough screening, receive detailed information about the study protocol, and provide informed consent before enrollment. Throughout the trial, researchers monitor outcomes closely, documenting healing times, implant integration, and any complications. Data collected contributes to scientific publications and regulatory approval processes.
Eligibility Criteria for Participation in Clinical Trials for Implants
Eligibility requirements for dental implant clinical trials vary depending on the specific study objectives and the stage of research. Common criteria include age restrictions, typically requiring participants to be adults over 18 years old with fully developed jawbones. Candidates must have adequate bone density and volume to support implant placement, though some trials specifically seek patients with bone loss to test advanced grafting techniques. General health status is carefully evaluated, as conditions like uncontrolled diabetes, active periodontal disease, or immune system disorders may disqualify candidates due to increased complication risks. Smoking status often affects eligibility, as tobacco use can impair healing and implant success rates. Some trials focus on specific patient populations, such as those missing single teeth versus multiple teeth, or individuals requiring immediate implant placement following extraction. Participants must commit to attending all scheduled follow-up appointments, which may span several months or years, and agree to detailed documentation including radiographs and clinical examinations. Exclusion criteria typically include pregnancy, recent radiation therapy to the head or neck, and use of medications that affect bone metabolism.
Potential Benefits and Considerations of Joining a Clinical Trial
Participating in a dental implant clinical trial offers several potential advantages alongside important considerations that candidates should carefully evaluate. Benefits often include access to innovative treatments not yet available to the general public, potentially representing the latest advances in implant technology or surgical methods. Many trials provide treatment at significantly reduced costs or completely free of charge, including the implant procedure, materials, and follow-up care. Participants receive exceptionally thorough monitoring from experienced dental professionals and researchers, often exceeding the level of attention provided in standard clinical practice. Contributing to scientific knowledge and helping future patients represents a meaningful aspect of participation for many individuals. However, considerations include the experimental nature of the treatment, which may carry unknown risks or prove less effective than established methods. Trial protocols impose specific requirements and restrictions, limiting flexibility in treatment timing or approach. Participants may be randomly assigned to receive either the experimental treatment or a standard procedure, without choosing their preference. Time commitments for appointments and assessments can be substantial, and some trials require travel to specific research centers. Potential side effects or complications must be carefully weighed, and participants should understand that they can withdraw from a trial at any time if concerns arise.
| Research Center | Trial Focus | Estimated Participation Cost |
|---|---|---|
| University of Milan Dental School | Novel titanium alloy implants | Free treatment, follow-up included |
| Rome Implantology Research Institute | Immediate loading protocols | €500-€1,000 reduced fee |
| Bologna Clinical Trials Center | Bone grafting with implant placement | Free grafting materials, standard implant fees apply |
| Turin Dental Innovation Lab | Digital planning and guided surgery | €300-€800 technology fee reduction |
Prices, rates, or cost estimates mentioned in this article are based on the latest available information but may change over time. Independent research is advised before making financial decisions.
Finding and Enrolling in Clinical Trials
Individuals interested in participating in dental implant clinical trials in Italy can explore several resources to identify suitable opportunities. University dental schools and teaching hospitals frequently conduct research and maintain registries of ongoing studies. The Italian Medicines Agency website provides information about registered clinical trials, though navigating medical terminology may require assistance from a dental professional. International clinical trial databases, including ClinicalTrials.gov and the EU Clinical Trials Register, list studies recruiting participants in Italy. Consulting with a general dentist or oral surgeon can provide guidance, as these professionals often have connections with research institutions and can assess whether a patient might be a suitable candidate. Patient advocacy organizations focused on oral health sometimes maintain information about research opportunities. Once a potential trial is identified, interested individuals typically undergo a preliminary screening, either by phone or in person, to determine basic eligibility before proceeding to comprehensive evaluation.
Regulatory Protections and Ethical Standards
Clinical trials in Italy operate under rigorous regulatory frameworks designed to protect participant welfare and ensure scientific validity. All studies must receive approval from ethics committees before enrolling patients, and researchers must demonstrate that potential benefits justify any risks. Informed consent procedures require that participants receive comprehensive information about the study purpose, procedures, potential risks and benefits, alternatives, and their right to withdraw without penalty. Data privacy protections ensure confidentiality of medical information collected during trials. Compensation policies vary, with some studies offering reimbursement for travel expenses or time, though payment for participation itself is generally limited to avoid undue influence. Adverse events must be promptly reported and investigated, and studies can be suspended if safety concerns emerge. Participants maintain the right to continue receiving appropriate dental care even if they withdraw from a trial, and researchers have obligations to provide or arrange necessary treatment if complications arise from study procedures.
Conclusion
Dental implant clinical trials in Italy represent an intersection of advanced dental care and scientific progress, offering selected individuals access to innovative treatments while contributing valuable data to the field of implantology. Understanding the trial process, eligibility requirements, and the balance of benefits and considerations enables informed decision-making for those considering participation. With strong regulatory protections and ethical standards in place, these studies continue to advance dental science while prioritizing patient safety and welfare.




